In recent analyses, OBELA observes an increase in the demand and use of land passenger transport. For this reason, we consider its electrification employing fuel batteries to be a point in favour of the environment and economy. However, there is a debate on whether to take advantage of the lithium present on land or invest in hydrogen (H2) production from natural gas or water. What is the best alternative to protect the environment and slow down climate change?
Hydrogen is the most abundant and lightest element on Earth, but its molecules do not naturally appear in isolation. Obtaining it requires greater technical and economic complexity, from a raw material utilizing an energy contribution. Currently, the primary source of H2 is natural gas and water. Electrolysis is the only process for extracting H2 that does not generate carbon dioxide (CO2), but separating H2 from the oxygen in the water consumes more energy than that produced by burning the H2. It is a highly flammable gas and requires safety systems that increase its production cost.
Three prevalent processes exist for the storage of the produced H2: the pressurization, the liquefaction, and the absorption in metallic hydrides. The most complex in its storage is that although H2 has one of the highest specific energy levels, it has one of the lowest levels of energy density. It requires very high pressures and extremely low temperatures (-253 ℃), which increase the cost and complexity of its pressurization.
Storage using metal hydrides (compounds where hydrogen combines with a metal) allows it to keep at relatively low pressures, with a high density without such extreme thermal demands. Although they degrade over time and are very sensitive to impurities, they offer more excellent safety and better characteristics for stationary systems, a convenient solution.
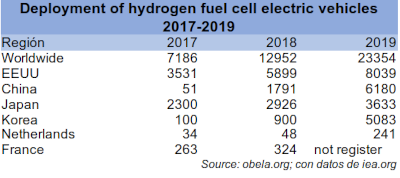
However, the recharging infrastructure for H2 batteries is still inefficient. The installation is more expensive and complicated than that of an electrical recharging network. Large transport vehicles such as trucks or trains are still far from using this type of battery for efficiency and energy consumption reasons. Currently, investment is being made in research to remove these limits.
Lithium, different from H2, can be found in specific deposits. The most important is the "Lithium Triangle," an area located on the border of Argentina, Bolivia, and Chile that concentrates more than 85% of the known reserves on the planet.
The battery industry explores different chemical combinations to improve the efficiency of its batteries. For example, the Chinese manufacturer Brighsun New Energy developed a lithium-sulfur battery, which promises to suppress dendritic growth, the main obstacle facing lithium-ion batteries. Dendrite is an internal cavity resulting from the battery's deterioration, which increases its flammability and reduces its safety.

Currently, treating these batteries as waste emits a lot of CO2, and only recover 50% of their components. For this reason, a proposal exists to reuse batteries that have lost part of their performance. When they are not useful as an energy reserve for electricity transport purposes any longer, they can go to emergency power systems in charging stations. There rapid charging and discharging are not so essential, and energy density is not a problem.
The extraction of lithium and natural gas (to obtain H2) generates severe impacts on the environment and many risks associated with the geology of the deposits: floods, landslides, fires, explosions, and the release of various pollutants. The countries with the largest reserves of lithium and hydrocarbons have the laxest laws on the subject.
The extent to which environmental law in each country will hold the battery producer accountable, from obtaining materials to managing waste, needs to be addressed. The expert working group on ESM (Environmentally Sound Management) within the Basel Convention is currently working on fact sheets to properly manage different types of waste. Although not yet finalised, an initial draft is available.
There is no such thing as a perfect source of energy. Environmentally, the best alternative is H2 from water, which does not generate any pollutants after burning. The problem lies in that it is not yet economically or energetically feasible. It takes twice as much electricity to produce the H2 for a car to travel theoretically 100km than that needed with lithium batteries, discounting that required for compression.










